Cl2o
Cl2O polar or not polar? Why? please explain!?!? ۔
Is the Cl2O molecule polar or non-polar and why? Any help is welcome!
There is a pole
Is Cl2o polar?
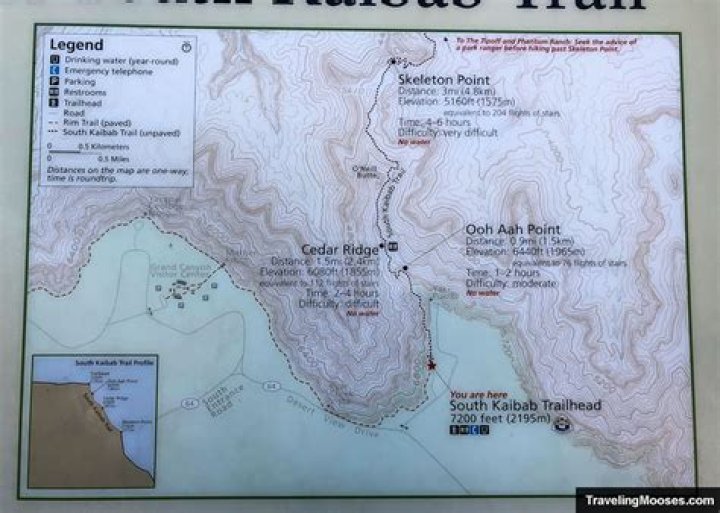
The molecular geometry of Cl2O is similar to that of H2O, except that the bond angle is 111 degrees. Polar bonds combine with curved molecular geometry to form polar molecules that have a slightly negative charge for oxygen and a slightly positive charge for chlorine atoms. The dupole moment is 0.78 D.
Cl2o
Cl2o
Cl2O polar or not polar? Why? please explain!?!? 3
Is the Cl2O molecule polar or non-polar and why? Any help is welcome!
The molecular geometry of Cl2O is similar to that of H2O, except that the bond angle is 111 degrees. Polar bonds combine with curved molecular geometry to form a polar molecule that has a slightly negative charge for oxygen and a slightly positive charge on the chlorine atom. The dupole moment is 0.78 d.